Turkey Pharmaceutical Market (Jan-June 2017 Report)
12 January 2018
We Participated in Expo Turkey by Qatar 2018 in Doha
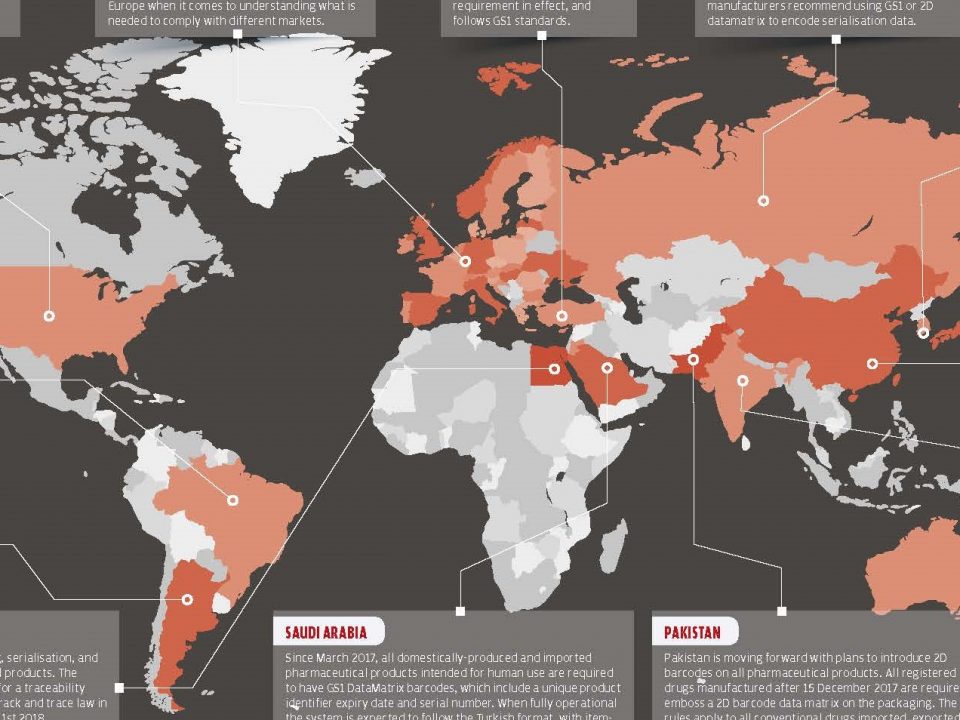
25 January 2018One of the most pressing challenges we face as a result of the globalization of health services over the years is to ensure the integrity and safety of the drug supply chain.
Fake and dangerous versions of drugs are illegally sold to international criminal organizations, weakening private and public investments made by threatening patient safety and public health. The scope of illegal multinational trade is very broad and complex.
Despite these known dangers to the public health, it has been seen that in the reports compiled by public and private stakeholders (including regulatory agencies, manufacturers and customs authorities); the sale, distribution and use of substandard, spurious, falsely labelled, falsified and counterfeit drugs (SSFFC) continues.
In the trade and distribution of SSFFC medicines there is a key systematic category in the international criminal organizations: SSFFC medicines identified in controlled and legal supply chains.
Specifically, we can define the legal supply chain as “a supply chain which starts from a producer/importer and end with an end user, also organized, inspected and licensed by the ministry of health or and approved organization”.
The introduction of counterfeit medicines into the supply chain is of great importance and requires focus on public health and policy research for three reasons.
- The identified fraudulent and illegal drug incidents have not been systematically assessed in detail when compared to illegal supply chains and unregulated markets.
- It has been determined that counterfeit and illegal drugs penetrate the legal supply chains that are weak and tightly controlled (such as malaria, cancer, HIV medicines, etc.). This problem has led to the need for more research into the future of counterfeit drugs in supply chains and supply chain safety issues.
- To prevent future fraud and counterfeiting incidents, decision-makers and government officials need to support studies and regulations in this regard.
It should also be noted that middle-income countries are critical to ensuring global drug safety. With the need to provide more detailed and up-to-date data, it has seen that middle-income countries are much more likely to engage in the production and distribution of counterfeit and illicit drugs. Like many developing countries, there is also the problem of counterfeit and illegal drugs being found within the legal supply chain in these countries. Because there aren’t established standards about this subject, counterfeit drugs are threatening global patient safety.
As the Organization for Economic Co-operation and Development (OECD) has stated, governments, companies and other interested parties must continue to collect and evaluate all the information necessary to effectively combat counterfeit drugs. Solutions should be produced using these assessments.